RESEARCH RESULTS
A double-blind, placebo-controlled clinical trial with 80 subjects showed that Coldbloc® works. The study tested the effectiveness of an antiviral Coldbloc® against the common cold and found a significant difference (P < 0.003) between Coldbloc® and the control. The study was modeled after Mossad’s research at the Cleveland Clinic, where they tested the efficacy of zinc lozenges for treating the common cold (Annals of Internal Medicine 125(2):81-88, 1996).
FAST RELIEF AVAILABLE!
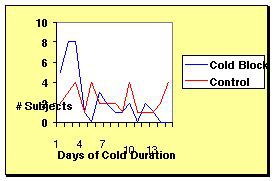
Coldbloc® decreased the duration of the common cold from 7.4 to 4.6 days.
Figure 1. Comparing the days of cold duration in the Coldbloc® group (4.6 days) versus the control group (7.4 days).
A double-blind, placebo-controlled clinical trial with 80 subjects showed that Coldbloc® works. The study tested the effectiveness of an antiviral Coldbloc® against the common cold and found a significant difference (P < 0.003) between Coldbloc® and the control. The study was modeled after Mossad’s research at the Cleveland Clinic, where they tested the efficacy of zinc lozenges for treating the common cold (Annals of Internal Medicine 125(2):81-88, 1996).
FAST RELIEF AVAILABLE!
Coldbloc® decreased the duration of the common cold from 7.4 to 4.6 days.
Figure 1. Comparing the days of cold duration in the Coldbloc® group (4.6 days) versus the control group (7.4 days).
SORE THROAT RELIEF
People were often surprised at the immediate relief of their sore throat symptom, and their speedy recovery.
DURATION REDUCED!
Coldbloc® blocked common cold symptoms within:
People were often surprised at the immediate relief of their sore throat symptom, and their speedy recovery.
DURATION REDUCED!
Coldbloc® blocked common cold symptoms within:
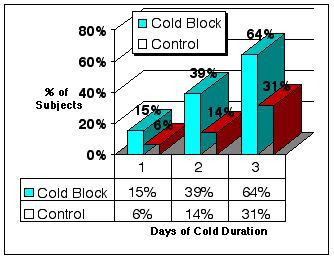
| 1 day | 15% of subjects | 6% in control |
| 2 days | 39% of subjects | 14% in control |
| 3 days | 64% of subjects | 31% in control |
Figure 2. Percent of subjects experiencing relief of cold symptoms after day 1, 2 and 3.
REDUCES DURATION OF LONG-LASTING COLDS
Even in cases where Coldbloc® did not appear to be "blocked" (3 days or less), Coldbloc® reduced the long duration of colds.
Number of people in the study with colds lasting 4 days or more:
Almost 3/4 (69%) of those in the control group.
Only 1/3 (36%) of those taking Coldbloc®.
One person indicated that Coldbloc® worked, but when questioned about his cold lasting one week, he said,"Yes, but my colds usually last three weeks."
Even in cases where Coldbloc® did not appear to be "blocked" (3 days or less), Coldbloc® reduced the long duration of colds.
Number of people in the study with colds lasting 4 days or more:
Almost 3/4 (69%) of those in the control group.
Only 1/3 (36%) of those taking Coldbloc®.
One person indicated that Coldbloc® worked, but when questioned about his cold lasting one week, he said,"Yes, but my colds usually last three weeks."